Why can’t the volume of gas become zero as its temperature decreases?
Volume of gas will never become zero. Let’s use the following model to explain.
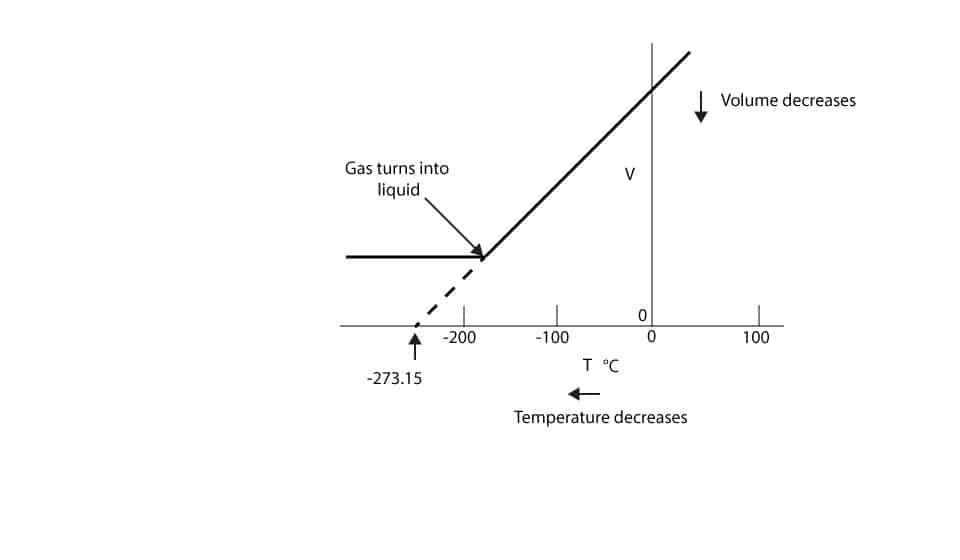
As the temperature of gas decreases it gets to a setting point where the gas turns into a liquid. Once the gas turns into a liquid, its volume change becomes so-so small that we can ignore. From the graph, you can see a leveling off of the volume at this point. As the temperature continues to decrease, the gas can turn into solid. From the graph, the volume of gas can never become zero. As you can see, it turns into a liquid at a volume value higher than zero. However, if we extend the original line (dotted part of line) to the temperature axis, you will notice that it crosses the temperature axis at -273.15 °C. If the gas had not turned into a liquid, the -273.15 °C will be the temperature the gas volume will become zero.
However, in real life, gas is matter and matter occupies space (volume), therefore, volume of gas can never become zero. But we can say that the temperature at -273.15 °C is the lowest temperature that can exist.
Can we design a temperature scale based on the lowest temperature possible?
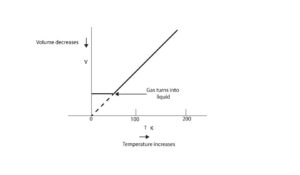
Yes, we can. This scale which was designed by Lord Kelvin is called the absolute temperature scale. Its unit is the kelvins (K). Let’s use the following graph to explain.
On the kelvins scale, the temperature starts at absolute zero, which is -273.15 °C on the Celsius scale. This absolute zero or sometimes called zero kelvins is written as 0 K.
Therefore, we can write this relationship between the two units as;
0 K = -273.15 °C
Since the Kelvins scale starts at 273.15 degrees below the Celsius scale, we can write that
°C + 273 = K. This just means that to convert temperature from degree Celsius to Kelvins, we must add 273 to the temperature value measured in °C.
For example, to convert 40 °C to Kelvins, we must add 273 (rounded) to it.
Thus, 40 °C + 273 = 313 K.
Notice that on the kelvin scale, we have moved the volume axis (vertical axis) to 273.15°C. Because we have done this, we can clearly see that the volume of the gas is directly proportional to the absolute temperature (T) of the gas. So as the temperature becomes zero the volume of gas becomes zero too. However, on the Celsius scale this is not the case. Thus, when the temperature becomes zero, the volume of gas does not equal zero.
Therefore, we are more correct when we say that the volume (T) of a gas is directly proportional to the absolute temperature (T) of the gas. This is the core reason we must convert temperature to kelvins when solving gas law problems.