How to use Lewis dot structures to explain covalent bonding
Let’s use the following models and octet rule to explain covalent bonding in simple molecules like: N2, CH2CH2 and CO32-.
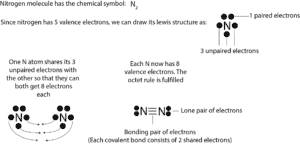
Nitrogen molecule Lewis dot diagram
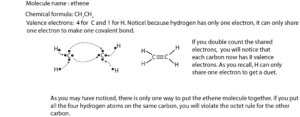
Ethene molecule Lewis dot diagram
Carbonate ion Lewis dot diagram

